New technology detects breast cancer relapse up to five years before symptoms appear using blood samples
A study led by Dr. Iñaki Comino-Méndez and Dr. Emilio Alba from the Hospital Universitario Virgen de la Victoria of Málaga/IBIMA and Dr. Ángel Guerrero-Zotano from the Instituto Valencia de Oncología (IVO), with funding from the pharmaceutical company Pfizer, shows that the liquid biopsy technology developed by Altum Sequencing can detect breast cancer relapse up to 68 months earlier than conventional clinical methods.

Altum Sequencing, a biotech company specializing in minimal residual disease (MRD) monitoring through liquid biopsy, has contributed as a technological partner to a pioneering study recently published in Breast Cancer Research. The project, coordinated by Dr. Iñaki Comino-Méndez from the Hospital Universitario Virgen de la Victoria of Málaga/IBIMA, demonstrates that circulating tumor DNA (ctDNA) analysis enables the early detection of recurrence in patients with hormone receptor-positive (HR+) breast cancer—the most common subtype among women.
Thanks to Altum’s technology, it is possible to detect one tumor cell among a million healthy ones from a simple blood sample, offering a powerful tool for post-treatment monitoring. In some cases, clinical relapse was detected up to 68 months before symptoms became evident through standard diagnostic tools, giving clinicians a valuable time advantage.
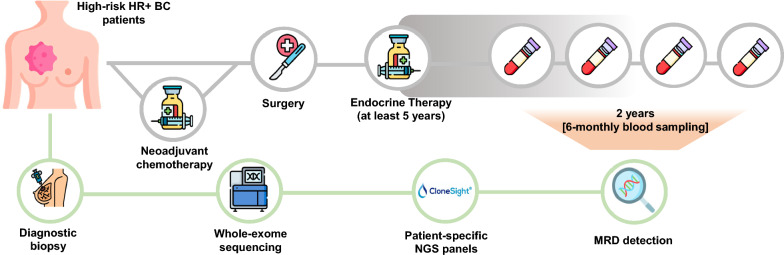
The methodology begins with an initial tumor biopsy to identify patient-specific mutations. Follow-up blood tests then screen for traces of those mutations in the bloodstream using next-generation sequencing (NGS). This personalized and minimally invasive approach reduces costs, avoids unnecessary treatments, and lowers the risk of false positives.
As Marina Planas, CEO of Altum Sequencing, explains, “Our goal is not to diagnose cancer, but to offer healthcare professionals a precise and sensitive tool to monitor disease progression after initial treatment.”
This breakthrough has potential far beyond breast cancer. The technology is adaptable to many types of solid tumors, and Altum is currently working on regulatory approvals in Europe and the U.S. to bring the test to hospitals worldwide.





