Four AseBio partners will participate in the first IPCEI in the health sector that will mobilize €1 billion in grants
The Major Project of Common European Interest 'Med4Cure' will enable member states to support a total of 14 projects from 13 companies that will result in more than 70 collaborations, as well as 11 associate members.

The European Commission has given the green light to an investment of 1 billion euros for “Med4Cure”, the fourth Major Project of Common European Interest (IPCEI), and the first in the health sector. Its mission is to “support research, innovation and the first industrial deployment of medical devices and innovative production processes for pharmaceuticals” in the European Union.
This project has been notified jointly by six EU Member States: Belgium, France, Hungary, Italy, Slovakia and Spain, which joined this project in the framework of the Recovery and Resilience Plan. “Med4Cure” finds its origin in the Joint Manifesto signed in March 2022 by 16 Member States for the implementation of a health IPCEI. A proposal that was born to support innovation in technologies related to drug production, addressing challenges such as antimicrobial resistance, rare diseases or emerging health threats, as well as the development of cell and gene therapies. The European Commission states that this IPCEI “will contribute significantly to the objectives of the European Health Union”.
Member States will provide up to €1 billion in public funding, and this project is expected to unlock an additional €5.9 billion in private investment. The IPCEI “Med4Cure” as a whole is expected to be completed by 2036 (timelines vary within this timeframe depending on the characteristics of each project and participating companies), and is expected to generate around 6,000 direct and indirect jobs.
In this regard, the European Commission has detailed that the research and development projects included in “Med4Cure” cover all the key stages of the pharmaceutical value chain: from the collection and study of cells, tissues and other samples, to the development of sustainable production technologies for innovative therapies, including personalized treatments or the application of advanced digital technologies.
“Med4Cure” encompasses a total of 14 projects from 13 companies that will result in more than 70 collaborations. The results will be widely shared by the participating companies with the European scientific community and industry, beyond the participating companies and countries.

These 14 projects are part of the broad ecosystem shaped by the IPCEI Med4Cure, which also involves 11 associated partners in four Member States (Belgium, the Netherlands, Poland and Spain). In addition, there are about 175 indirect partners, companies or research organizations that will benefit from the various dissemination activities of the ICEPEI Med4Cure, as shown in the following image:
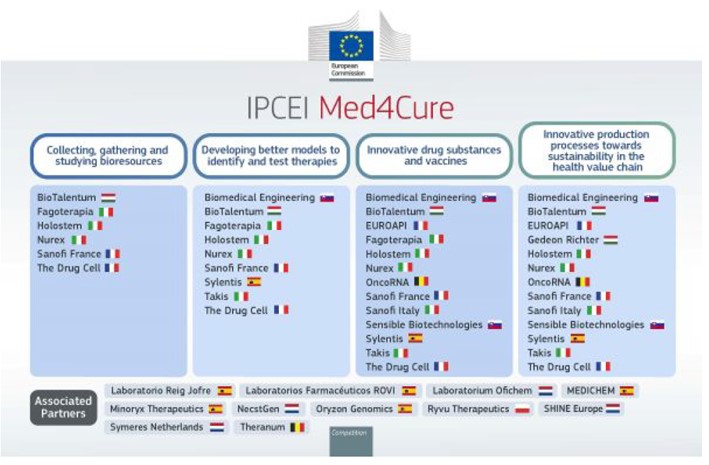
Among the 13 companies that make up the IPCEI “Med4Cure” is Sylentis, a pioneer in the development of novel drugs based on RNA interference (RNAi) technology, which will participate in the areas of the project aimed at developing better models for identifying and testing therapies, development of new drugs and vaccines, and innovative production processes that improve sustainability in the healthcare value chain.
In addition, five additional Spanish companies will be associate members of IPCEI, including three AseBio partners; Reig Jofre, Minoryx Therapeutics, ROVI, Oryzon Genomics, as well as Laboratorios ROVI and MEDICHEM.
AseBio congratulates the Spanish companies participating in the IPCEI and hopes that their participation will contribute to boost their activities and impact on the Spanish biotechnology sector.





